Innovative Treatment for a Pipeline of Inflammatory Indications
CytoAgents is advancing CTO1681 for multiple indications where dysregulated cytokine signaling plays a central role in disease pathogenesis. Therapeutic indications include pulmonary arterial hypertension (PAH), asthma, atopic dermatitis (AD), and inflammation within the tumor microenvironment.
Our Vision
Immune hyperactivation and dysregulation are key drivers of disease pathogenesis and complications across oncology and inflammatory diseases, specifically in the tumor microenvironment, as well as in asthma, COPD, and atopic dermatitis. Hyperactivation of the immune system limits efficacy of cell therapies and causes life threatening treatment-related toxicities. Greater than 60% of acute exacerbations in asthma and COPD are driven by viral infections that further drive prostaglandin and prostacyclin-mediated inflammation. Similarly, greater than 90% of atopic dermatitis patients are colonized with S. Aureus and many are complicated with concomitant viral/fungal infections that drive disease flares and progression.
CTO1681 aims to solve this problem by providing a safe, effective and orally bioavailable immunomodulator that will help overcome the inflammation driving illness. CTO1681 is a novel, steroid sparing inhibitor of prostaglandin/prostacyclin-mediated inflammation. Our mission is to enable greater accessibility, lower costs, increased clinical adoption, and most importantly better outcomes for all patients.
CytoAgents: Our Journey
2018
FOUNDED WITH A FOCUS ON CYTOKINE RELEASE SYNDROME (CRS)
2019 – 2021
BUILT THE FOUNDATION
Advanced our proprietary prostacyclin platform targeting immune activation
and dysregulation2022 – NOW
ADVANCING OUR LEAD PROGRAM
Advanced CTO1681 in oncology and CAR-T related CRS, currently enrolling our
Phase 1b/2a Clinical TrialLATE 2025
EXPANDED OUR PIPELINE
Expanded into Pulmonary Arterial Hypertension (PAH), Asthma, and Atopic
Dermatitis — a broader “pipeline-in-a-pill” strategyTHE ROAD AHEAD
BEST-IN-CLASS ORAL ANALOGUE
Advancing a best-in-class oral prostacyclin analogue into Phase 2/3 clinical
trials across multiple high-need indications
Pipeline of Inflammatory Indications
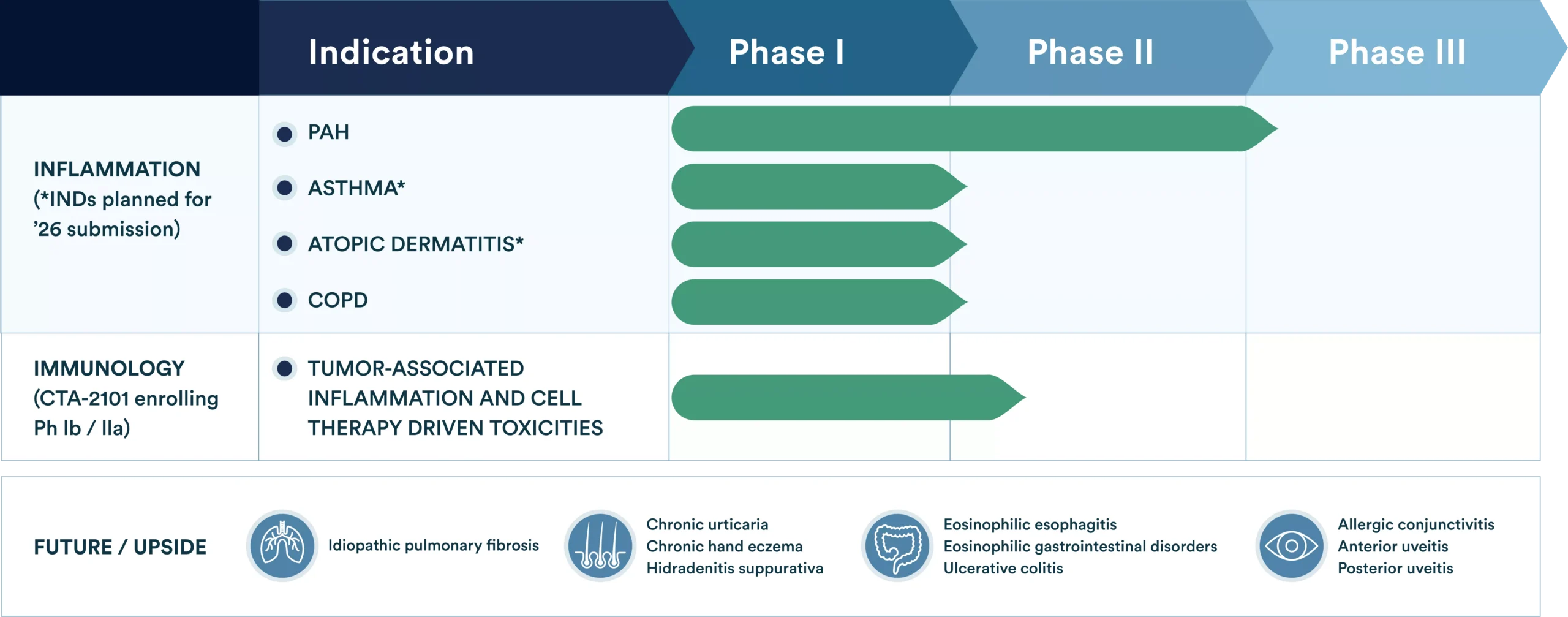

Idiopathic Pulmonary Fibrosis
Chronic urticaria
Chronic hand eczema
Hidradenitis suppurativa
Eosinophilic esophagitis
Eosinophilic gastrointestinal disorders
Ulcerative colitis
Allergic conjunctivitis
Anterior uveitis
Posterior uveitis
How CTO1681 Works
CTO1681 is a novel prostacyclin/prostaglandin analogue designed to modulate both vascular and inflammatory pathways through selective receptor signaling. Through activation of the IP receptor (prostacyclin pathway), CTO1681 has the potential to deliver therapeutic benefit in pulmonary arterial hypertension (PAH).
In addition, selective signaling through the EP4, EP3, and EP1 receptors (prostaglandin pathways) while sparing EP2 receptor signaling helps modulate immune activation and inflammatory responses, supporting potential benefit across a broad range of inflammatory diseases including asthma, and atopic dermatitis.
By targeting upstream inflammatory signaling while preserving normal immune function, CTO1681 is designed to provide a differentiated, steroid-sparing approach to chronic inflammatory disease management.
- Oral, small molecule therapy with flexible delivery options
- Selective prostacyclin/prostaglandin modulation through the IP, EP4, EP3, EP1 receptors while sparing EP2 receptor activity.
- NDA Enabling safety package
- Efficacy has been de-risked in PAH
- Steroid-sparing approach designed to reduce reliance on chronic steroid use
- Non-JAK with more favorable safety profile
CTO1681 Clinical Development
CAR T-Cell and TCE Therapy-Related Inflammation
CytoAgents is conducting its Phase 1b/2a clinical trial aimed at reducing inflammation in patients receiving CAR T-Cell Therapy in oncology. CAR T therapies face a dual challenge in oncology indications. First, inflammation in the tumor microenvironment may limit the CAR T from properly expanding and effectively killing cancer cells. Second, immunotherapies such as CAR T and TCE therapy trigger powerful immune responses to fight disease, which often induces cytokine driven toxicities such as CRS and ICANS. CTO1681’s ability to safely modulate the cytokine response while keeping the immune system functionally intact makes it an ideal solution in this setting.


CytoAgents is conducting its Phase 1b/2a clinical trial aimed at reducing inflammation in patients receiving CAR T-Cell Therapy in oncology. CAR T therapies face a dual challenge in oncology indications. First, inflammation in the tumor microenvironment may limit the CAR T from properly expanding and effectively killing cancer cells. Second, immunotherapies such as CAR T and TCE therapy trigger powerful immune responses to fight disease, which often induces cytokine driven toxicities such as CRS and ICANS. CTO1681’s ability to safely modulate the cytokine response while keeping the immune system functionally intact makes it an ideal solution in this setting.

CTO1681:
Prostaglandin Inhibitor reduces PGE2 Signaling and modulates 20+ key cytokines
Our Science
MODULATING INFLAMMATION & CYTOKINE AMPLIFICATION
Many illnesses develop when unchecked cytokine production leads to a cycle of inflammation. CTO1681, an oral prostaglandin inhibitor treats the underlying causes of inflammation by blocking the NF-kB and PGE2 signaling pathways that play a critical role in activating and regulating inflammatory immune cells. By interrupting these signaling pathways CTO1681 effectively blocks cytokine amplification, downregulating cytokine production while leaving the immune system functionally intact.
Partners & Investors
CytoAgents has made tremendous progress in controlling cytokine driven inflammation. We have successfully completed our initial Phase 1 Human Clinical Trial and are currently enrolling a Phase 1b/2a Clinical Trial in patients receiving CAR T-cell therapy at risk for cytokine driven overwhelming inflammation.
Join us as we transform inflammation through a novel approach that allows for prevention as well as treatment.
Contact Us
